|
Page 1, 2
I have important reasons for making robust efforts to detect insulin dysregulation and improve insulin homeostasis in every patient with cancer under my care. My primary reasons are the following: (1) Energetically, both cancer and insulin dysregulation are rooted in mitochondrial dysfunction1-5; (2) Malignant cells carry a larger number of insulin receptors in their cell membranes than do non-malignant cells6, which they employ to meet their larger needs of fuel for cellular growth and spread; (3) Disease processes of cancer and insulin dysfunction fan each others' fires by diverse mechanisms; (4) The two diseases often share co-morbidities; (5) Excess insulin is pro-fermentation and proinflammatory, the two major mechanisms of malignant cell proliferation; and (6) Insulin is a direct growth factor for cancer.
 In my patients with cancer, I find some degree of hyperinsulinism in nearly all patients, usually beginning within months of the onslaught of fears and doubts, real or imagined, concerning cancer diagnosis as well as the toxicity and efficacy of treatment options. In this paper, I present the findings of insulin studies in five cases of cancer, three involving breast cancer in women and two involving prostate cancer. In my patients with cancer, I find some degree of hyperinsulinism in nearly all patients, usually beginning within months of the onslaught of fears and doubts, real or imagined, concerning cancer diagnosis as well as the toxicity and efficacy of treatment options. In this paper, I present the findings of insulin studies in five cases of cancer, three involving breast cancer in women and two involving prostate cancer.
In my integrative clinical work, I include assessment of insulin homeostasis as a part of my comprehensive evaluation of all patients with chronic illness with clinical and laboratory methods. I may point out here that I now read insulin and glucose profiles prepared during this work-up as I read surgical pathology slides during 29 years of my work as a hospital pathologist, always inclusive of all clinical and laboratory findings. Wide variations in insulin responses to a glucose challenge among profiles of individual are the rule, not an exception – as is the case in diagnostic histopathology. My patients have taught me to do so for all patients with chronic gut-related, allergic, immune-inflammatory, endothelial-cardiovascular, digestive-absorptive, hepatic, neurologic, metabolic and non-metabolic disorders. I do so in view of the enormous range of serious non-metabolic consequences of hyperinsulinism and kaleidoscopic interweaving of oxygen and insulin signaling at all levels in nearly all body organs of the body.7-9 Study of insulin homeostasis in this larger context is far more revealing and clinically valuable than is possible with a merely glycemic focus.
Below is some text from my recent comments on the subject entitled Breast Cancer Driver Mutations e-published by the journal Nature10 (with re-numbered references and tabular format for presenting insulin data concerning three cases of breast cancer):
The work of Nik-Zainal et al.11 enlightens physicians who treat cancer with an integrative model in two ways: (1) it vastly expands the knowledge of breast cancer genome which is expected to yield significant clinical benefits in coming years; and (2) it underscores the importance of addressing non-genetic elements in treating breast cancer. The finding of 93 protein-coding breast cancer genes carrying probable driver mutations notably extends the range beyond the limits of BRCA 1 and BRCA 2 mutations. The repertoire of breast cancer genes and mutational processes revealed in the study further underscores the need for integrating new treatments based on advances in tumor genomics with those made with dietary, metabolic, environmental, and self-regulatory approaches to counter tumor-related immunosuppression. Specific concerns here are: (1) diagnosis-related anger and fear; (2) cumulative burdens of cancer biology and comorbidities; (3) imagined and real concerns about serious adverse effects of treatment options; and (4) perceived and actual probabilities of treatment failure.
To address cancer-related immuno-suppression from diverse causes, the author focuses on oxygen signaling12,13 and insulin homeostasis.13,14 In 1995, a holistic-integrative model of cancer control based on accelerated oxidative injury was proposed as the common denominator in elements that sustain and promote proliferation of tumor cells.8 In 2001, the oxidative hypothesis was expanded to the dysox model of cancer with the following words: cancer is the destructive behavior of cells incited and perpetuated by many factors that cumulatively lead to anomalous oxygen signaling.15 In 2009, focus on issues of insulin homeostasis led to proposing an oxygen model of hyperinsulinism.16 and described a hyperinsulinism modification plan.17
Breast cancer cells carry a larger complement of insulin receptors than non-neoplastic mammary gland cells.6 This may be expected to add to disruption of insulin homeostasis caused by dietary, metabolic, and other factors mentioned earlier and indeed might become a clinically important metabolic concern. The author investigated this risk by measuring insulin responses to a 75-gram glucose challenge with blood samples drawn at fasting and 1-hour, 2-hour, and 3-hours in patients with a variety of malignant neoplasms. The following sets of blood insulin and glucose data concerning three women with unimpaired glucose tolerance and breast cancer treated with chemotherapy and surgery illustrate my main points: (1) how insulin can rise with treatment (Table 1, cases 1 and 2); and (2) how insulin levels can fall with an integrative insulin modification plan following treatment (Table 1, case 3). The insulin profile of a healthy subject with unimpaired glucose tolerance is included as a control. Insulin and glucose concentrations in 3-hour insulin and glucose profiles given below are expressed in uIU/ml and mg/mL respectively….
The author is one of the growing community of physicians who treat cancer and recognize the deep commitment of scientists to advancing science for controlling cancer. They also acknowledge that the scientists' diligence, persistence, and endurance is not being matched by that of clinicians. The patients often pay a heavy price when relevant complementary immunity-bolstering nutritional, metabolic, environmental, and self-regulatory measures are withheld from.
Limited clinical information about three women with breast cancer referred to in the preceding excerpt is included in that section. Table 1 displays their insulin and glucose profiles. Compared with blood insulin concentrations of the control subject, insulin levels are higher in all three. Note the rise in post-chemotherapy profiles in Cases 1 and 2, and a drop in the peak insulin level from 96 uIU/mL to 23 uIU/mL in response to the diet and detox measures in Case 3.
(If you prefer, all three tables are in this .pdf.)
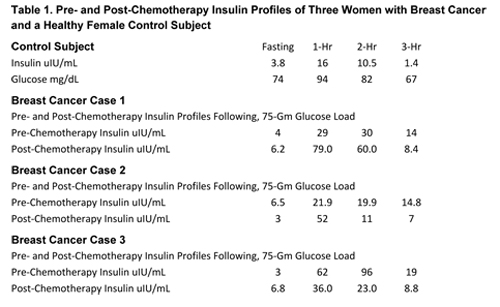
No clear inference can be drawn from the findings in just three cases of breast cancer, nor can any be drawn from just two cases of prostate cancer presented in Tables 2 and 3. However, when considered together, insulin responses observed in all five cases do point to a trend which, if validated by additional data and seen in light of established adverse effects of hyperinsulinism,18,19 might prove to be clinically significant.
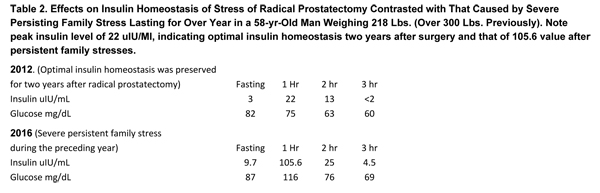
Notwithstanding the study sample of only two cases of prostate cancer, the case studies of both men are quite revealing in the context of glycemic and insulin responses to changing clinical contexts. Table 2 presents the case of a patient with prostate cancer. Insulin profiles of Patient #1, obtained two and six years after radical prostatectomy, provide an illuminating contrast between the insulin responses to stress of radical surgery and severe stress of family circumstances which developed. The profile obtained two years after surgery (in 2012) unexpectedly reveals a near-perfect insulin homeostasis (in the evolutionary sense that very low insulin levels are accompanied by unimpaired glucose tolerance), whereas family stresses evoked a strong insulin response, the peak insulin level rising more than fivefold (from 22 uIU/mL to 105 uIU/mL).
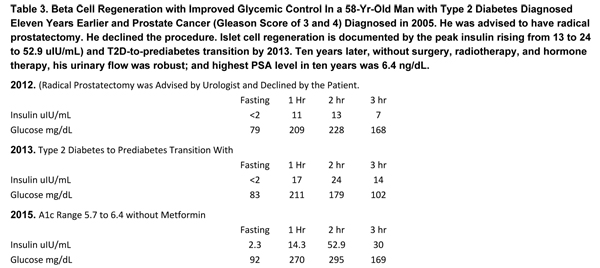
The second case of prostate cancer (Table 3) dramatically documents the expected greater clinical benefits of integrative management of co-morbidities. Specifically, twelve years after the initial diagnosis of prostate cancer, his prostate lesion is kept under satisfactory control with integrative therapies that did not include surgery, radiotherapy, or hormone therapies. His urinary flow is robust and his highest PSA level was 6.4 ng/dL.
Evidence of beta cell regeneration with superior glycemic control is drawn from (a) peak insulin level rising from 13 to 24 to 52.9 uIU/mL); and (b) T2D-to-prediabetes transition seen in the insulin profile obtained in 2013.
The author's main point: It can be reasonably argued that (a) improvement in insulin homeostasis with integrative therapies helped achieve the goal of satisfactory tumor-control without surgery, radiotherapy, and hormone therapies; and (b) control of the malignant lesion without surgery, radiotherapy, and hormone therapies increased the probability of improving the status of insulin homeostasis.
Page 1, 2
|
![]()
![]()
![]()
![]()








