|
Page 1, 2
An excellent example of true therapeutic synergy was discovered in the earlier days of my IV research (under the NIH-funded Bastyr Integrative Oncology Research Center) in the combination therapy using Poly-MVA and dichloroacetate (DCA) for both IV and oral use. We will describe the basis of the synergy and the initial case series that was followed.
Before discussing the combined metabolic therapy we developed, it would be useful to discuss the components separately in regard to their use in cancer. Those components are DCA, retinol, hyperbaric oxygen, and finally Poly-MVA versus alpha lipoic acid.
DCA
DCA is a relatively small molecule, which has been used as treatment for lactic acidosis. It inhibits lactate formation and releases pyruvate dehydrogenase kinase from negative regulation, thus promoting pyruvate entry into the TCA cycle. This increases oxygen consumption and reactive oxygen species (ROS) formation while glycolysis and lactate formation are repressed.1 Non-cancerous human cells prefer this aerobic pathway for energy formation via the electron transport chain (ETC) use. Cancerous cells experience the Warburg Effect where most glucose is converted to lactate regardless of oxygen availability.2 Forcing a cancerous cell into TCA/ETC use thereby increases ROS formation and oxygen consumption.3
 Townsend Letter provides a platform for those examining and reporting on functional and integrative medicine. Please support these independent voices. Townsend Letter provides a platform for those examining and reporting on functional and integrative medicine. Please support these independent voices. |
Cancers targeted in the published data included glioblastoma and are targeted due to their reliance on glucose metabolism, as well as the ability of DCA to cross the blood brain barrier.4 Other cancer cell types which have shown sensitivity are breast, prostate, colorectal, pancreatic and endometrial cancers.5
The most common toxicity is a dose dependent reversible peripheral neuropathy. Other reactions appear to be mediated by a slowing of glutathione activity via the GSTz pathway: "From the Abstract: Dichloroacetate (DCA) inhibits its own metabolism and is converted to glyoxylate by glutathione S-transferase zeta (GSTz). … Moreover, DCA-induced inhibition of tyrosine catabolism may account for the toxicity of this xenobiotic in humans and other species."6 As, clinically, most toxicity effects appear to be mitigated either by slowing infusion, adding glutathione and nutrient support, or both, the use of such additional measures is indicated.
Retinoids
Retinoids (i.e., vitamin A, all-trans retinoic acid, and related signaling molecules) induce the differentiation of various types of stem cells. Nuclear retinoic acid receptors mediate most but not all the effects of retinoids. Retinoid signaling is often compromised early in carcinogenesis, which suggests that a reduction in retinoid signaling may be required for tumor development. Retinoids interact with other signaling pathways, including estrogen signaling in breast cancer. Retinoids are used to treat cancer, in part because of their ability to induce differentiation and arrest proliferation. "Retinoid research benefits both cancer prevention and cancer treatment."7 Retinoic acid has been investigated extensively for its use in treating different forms of cancer not only in prevention but also in treatment: "Under normal circumstances in the body, retinoic acid does preventive work against cancer formation. After cancer formation, retinoic acid becomes an attacker to cancer cells, one that blocks their growth and division and also triggers their differentiation and death through specific pathways."8 Nuclear retinoic acid receptors mediate most but not all the effects of retinoids. Retinoid signaling is often compromised early in carcinogenesis, which suggests that a reduction in retinoid signaling may be required for tumor development. Retinoids interact with other signaling pathways, including estrogen signaling in breast cancer. Retinoids are used to treat cancer, in part because of their ability to induce differentiation and arrest proliferation. "Retinoid research benefits both cancer prevention and cancer treatment."7 Retinoic acid has been investigated extensively for its use in treating different forms of cancer not only in prevention but also in treatment: "Under normal circumstances in the body, retinoic acid does preventive work against cancer formation. After cancer formation, retinoic acid becomes an attacker to cancer cells, one that blocks their growth and division and also triggers their differentiation and death through specific pathways."8
Hyperbaric Oxygen Therapy (HBOT)
HBOT is widely used as an adjunctive treatment for various pathological states, predominantly related to hypoxic and/or ischemic conditions. It also holds promise as an approach to overcoming the problem of oxygen deficiency in the poorly oxygenated regions of the neoplastic tissue. Occurrence of local hypoxia within the central areas of solid tumors is one of the major issues contributing to ineffective medical treatment. HBOT alone offers limited curative effect and is typically not used as monotherapy. In most oncology settings HBOT is used as a treatment along with other therapeutic modalities. An excellent review published in 2016 by Ostrowski et.al discusses the recent data regarding safety, efficacy and potential uses of HBOT in oncology and is a highly recommended resource.9
Poly-MVA
Poly-MVA is a redox molecule that facilitates energy charge transfer at the cellular level with regards to the mitochondrial respiratory chain; it can therefore protect (by accepting a radical electron) and provide energy (by increasing mitochondrial activity). Poly-MVA (also referred to as Pd-LA) is a polymer (liquid crystal) rather than a single molecule. It differs from free radical scavengers (e.g. alpha-lipoic acid) since there is no free lipoic acid or nutrients as they are irreversibly bound together in a polymer resulting in a molecule that is both fat- and water-soluble. Therefore, the polymer provides a unified redox reaction. In summary it is an extremely effective energy transferring molecule. Poly-MVA has been shown to be neuroprotective and helpful in supporting the mitochondrial complex.10-13
One reason to consider Poly-MVA in a combined therapy is mitochondrial support as this has the potential to aid metabolic therapies by strengthening normal cells and potentially weakening cancer cell metabolism. A study14 looking at the effects of Poly-MVA on mitochondrial dynamics revealed:
The level of GSH was also significantly improved and the level of lipid peroxidation was decreased significantly (p<0.05) by POLY-MVA. The results indicate that POLY-MVA is effective to protect the age-linked decline of myocardial mitochondrial antioxidant status. The findings suggest the use of this formulation against myocardial aging.
Alpha Lipoic Acid (ALA)
ALA has been used for many years in various cancer therapies. In a 2012 paper,15 the authors looked at neuroblastoma cells and potential for metabolic effect from both DCA and ALA. Their conclusions are revealing as to one potential mechanism by which ALA can have an anti-cancer effect:
These data suggests that LPA [ALA] can reduce (1) cell viability/proliferation, (2) uptake of [18F]-FDG and (3) lactate production and increase apoptosis in all investigated cell lines. In contrast, DCA was almost ineffective. In the mouse xenograft model with s.c. SkBr3 cells, daily treatment with LPA retarded tumor progression. Therefore, LPA seems to be a promising compound for cancer treatment.
The question arises: "Why consider Poly-MVA over ALA in a metabolic therapy?" In the past, our experience was to use ALA with DCA as well as other support nutrients. This strategy was typically able to mitigate the DCA side effects. The down sides of the combination therapy however were that it required multiple supplements and, in the IV form, required slower dose escalation of ALA due to potential side effects. Additionally, while ALA has some cancer metabolism effect, based on the data presented elsewhere in this paper and multiple patient responses, ALA did not have either the same level of synergy with DCA (as Poly-MVA) nor the potential metabolic benefits of Poly-MVA in combination with DCA. This led us to choose Poly-MVA as the neuroprotective and mitochondrial agent over ALA and support nutrients.
Why Combined Therapies?
The potential side effects of DCA (which can include neurological toxicity) and a deeper look into the mechanism by which DCA works led myself and Dr. Gurdev Parmar to postulate that Poly-MVA and DCA could have two areas of synergy if used together: One being a mutual anti-cancer benefit and the other to improve the safety and tolerance of the DCA. The first step after looking at the chemistry was to have a cell line study done to see if the synergy we saw "on paper" translated to cancer cell death. The short story is that both Poly-MVA and DCA had tumor kill but together they had additive benefit, and less DCA could be used with the same tumor kill.16 This was the best of both worlds with regard to a potential synergistic combination.
As we all know, what works on paper does not always work in the petri dish, and what works there doesn't always translate to animal or human use. Because of this I (from prior use of DCA and Poly-MVA) knew how to administer both agents safely so I knew that I could provide the therapy without any risk other than risks common to other IV therapies. I did however have to select a group of people with advanced cancer that had failed all therapies (standard oncology therapies and natural therapies) and consented to this as a trial of unknown outcome (in oncology research a "salvage therapy trial").
We began with a small group of patients, acquiring them one by one based on the above criteria, and over the course of two years implemented the therapy. The original case series is summarized in the table (left) and was part of the original study, but has not been published separately since reported (in the context of the whole trial outcomes) at the Society of Integrative Oncology.17 For reference, many times things discovered in studies take years to be published, if at all.
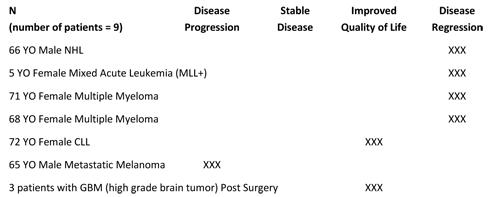
The basis of the therapy is outlined in the DCA and Poly-MVA sections above, but essentially the goal is to attack the cancer cell where it is weakest via its unique (but impaired) metabolism relative to normal human cells.18-24 I and colleagues have used this therapy, and the newer versions of it, many times in the years since and have had similar results. Of course, nothing works for everyone, but this combination has certainly improved overall cancer outcomes in our clinical experience. It should be noted the original protocol involved dietary changes (to a low carbohydrate or ketogenic diet) and a small group of oral supplements.
It should be of significant note that the only difference in the "disease regression" group and the other groups in the table above was that the "disease regression" group were the most stringent on their diet changes. This became a reason to increase the focus on the dietary portion of the intervention in future patients.
I had never seen these results when using DCA alone, so in my opinion the synergy seen is the petri dish worked in humans. Additionally, the rate of side effects from the DCA was drastically reduced, such that nobody since has had to drop out of the therapy due to DCA-related side effects. Overall, this is one of the truly big advances in integrative cancer therapies in the past twenty years.
In moving this therapy forward, I encountered the work that Dominic D'Agostino (of the University Of South Florida School of Medicine) was doing on metabolic oncology therapies in 2014 at the International Hyperbaric Medical Association.25 His work with animals and mine in humans had many crossover points. The main addition when I looked at both protocols was to combine hyperbaric oxygen therapy (HBOT) and exogenous ketones with my protocol.
Since 2014, we have treated many Stage-4 cancer patients with the combined metabolic therapy mentioned below. It has been safe and overall very effective in slowing disease, causing regression or stabilizing advanced cancer.
I believe based on the experiences of the past seven years with this evolving therapy that it holds a significant place as an effective intervention in advanced cancers. And while nothing certainly works universally in advanced cancer, a combined metabolic protocol should be considered as a potential therapy in all cases.
Page 1, 2
|
![]()
![]()
![]()
![]()







