Page 1, 2, 3
The test results showed strong hypothalamic-pituitary response to the small clomiphene dose of 25 mg every other day: LH and FSH values were both high (Table 2). In response to the elevated gonadotrophic hormones, his total testosterone – 288 ng/dL – was only 72% of the previous therapeutic value. However, this amount was greater than before he started TRT… and higher even than one of his earlier levels while using TRT. Most importantly, his free testosterone (10.1 pg/mL) was normal and the highest it had been in years!
Had his testicles atrophied during TRT, or did Case 2 have combined-type hypogonadism – or does clomiphene suppress his hepatic SHBG synthesis? Although resuming a small dose of TRT was discussed, Case 2 declined; he had improved so much while waiting for the test results that he opted to continue without any: he felt confident that his testicles would increasingly respond to the gonadotropins.
His confidence was validated on the next set of tests in July 2016. With LH and FSH values still a bit elevated, both total and free testosterone values were normal (Table 2). The ratio of tTEST/DHT is again normal at 12.3, and all his androgens, including DHT, are expected to continue to increase as LH and FSH "rehab" his gonads. Currently, he pays $25 monthly for clomiphene, compared with $85 to $150 monthly for compounded testosterone from various pharmacies.
5alpha-Reductase: Part 2: Further Actions
There are other significant roles for 5α-R beyond sex hormone metabolism and prostate cancer. As above, its substrates include other steroid hormones, their precursors and even metabolites ("breakdown products"), and endocrine modification is a key 5α-reductase function.7
Aldosterone, when 5α-reduced, becomes even more potent in signaling the kidney to retain salt and raise our blood volume (and blood pressure). In contrast to its effects on this hormone and testosterone, 5a-R significantly reduces the potency of cortisol. Ultimately, the same goal is achieved, though: signaling is diversified at each hormone's single receptor.62,63
5α-reductase takes progesterone remarkably farther, with very different consequences for men and women (the subject of the companion article). It is well known that sex hormones are "neutrally active": They alter our brain function, including our affect, thoughts, and behavior; in addition, they are neuroprotective.64,65 Sex hormones made in the body easily enter the brain and make us act, well, hormonal.
5α-R Makes Neurosteroids
Yet some steroid hormones influencing our brain are made within the brain itself, which like the skin (its embryological progenitor) possesses all the enzymes needed to make any steroid hormone de novo from cholesterol.66 These products are termed neurosteroids, and they are not controlled by hypothalamic-pituitary regulation.67 Progesterone enters this "realm" of neurosteroids via 5α-R and an acquiescent companion enzyme, 3α-hydroxysteroid dehydrogenase (3α-HSD; also called 3α-oxidoreductase to confuse casual readers).
In two steps, of which 5α-R is rate-limiting, these enzymes convert progesterone within the brain to the polysyllabic neurosteroid 3α,5α-tetrahydro-progesterone (also called allopregnanolone, or simply "ALLO") – see Figure 2.7,66,67 An aldosterone precursor, deoxycorticosterone is converted by the same enzymes to a neurosteroid with similar effects called 3α,5α-tetrahydro-deoxycortisone (THDOC). We shall focus on ALLO.
Figure 2
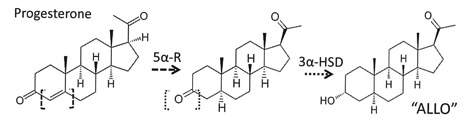
This key enzyme 5α-R is found together with 3α-HSD in neurons and glial cells within brain regions critical for mood, emotion, and sexual activity – no coincidence.67,68 Their neurosteroid products, ALLO and THDOC, travel via autocrine, paracrine, or transmembrane diffusion to influence the neuronal GABAA-receptor (GABAA-R).69 It is this receptor upon which the neurotransmitter GABA, benzodiazepines, barbiturates, and ethanol all exert their similar – and synergistic – effects.70
In binding to the GABAA-R, ALLO and THDOC potently enhance GABAergic inhibitory effects and influence the contrasting balance between GABA and glutamatergic (stimulatory) transmission.67 These neurosteroid effects are regulated and modified by the actions of local enzyme production and degradation, counteracting neurosteroids (e.g., 3β-THP) and the opposing effects of neuroactive steroids from the body.65,66,68,72,73
Under acute stress, effects of the GABAA-R inhibit hypothalamic corticotrophin-releasing factor (CRF) release, causing reduced anxiety and stress-behaviors. In rats and mice, infusions of the neurosteroids ALLO and THDOC into the brain enhance this GABAA-R activity, adding to its calming, protective effect.74-76 Neurosteroids – especially ALLO – are thought to help adjust, even "program," the HPA-axis response to stress.66
With evidence that infusing ALLO into animal brains reduces anxiety and stress-behaviors – and even mitigate seizures – it is logical to ask whether deficient ALLO is related to human mood disorders.67 Evidence shows that it can be. Circumstantially, we know that drugs used as antidepressants enhance neurosteroid levels, including clonazepine and olanzapine, lithium, fluoxetine, and some atypical anticonvulsants (valproate, lamotrigine, and carbamazepine ).77
More directly, low ALLO is linked to dysphoria in a study by Kilts et al. from the Durham VA Medical Center. Observing that nearly half of returning veterans experience persistent pain postdeployment, they studied the first 90 male US military veterans to have blood drawn upon entering the Mental Illness Research, Education and Clinical Centers (MIRECC) Registry. They found low ALLO blood levels significantly associated with increased pain perception.20
Why do these men have low ALLO? Possible explanations are a lack of 5a-reductase activity or, more likely, deficient progesterone as substrate. ALLO is reported to match progesterone levels throughout women's cycles.78,79 The men's chronic stress may be related to low progesterone. Progesterone is also the precursor of cortisol, the most important steroid and arguably the central hormone made in the stress response.80 It is popularly argued that progesterone can be depleted by producing cortisol in chronic stress states, known as "progesterone steal." Personal experience with patients supports this idea, though there is a paucity of published literature.
An intriguing possibility is raised: if treatment for ALLO deficiency were to be offered, supplying progesterone would be a physiological method. This has been done successfully at Yale, in a study of people stressed by cocaine addiction.81 It may not even be necessary to determine whether the veterans' pain was due to low ALLO or the lack of progesterone's valued neuroprotective effect.82 The authors' clinical experience suggests exercising caution in dosing men; however, as progesterone blood levels greater than 2 ng/mL can lead to increased abdominal adiposity and some gynecomastia.
Adding another layer of complexity, GABAA-receptor sensitivity to neurosteroids (and drugs) can be adaptively altered in response to prolonged changes in neurosteroid levels.83,84 In response to different types and amounts of stimulation, the receptors' subunit composition, phosphorylation state, and indeed total population can be altered – and then may return to their original state as circumstances allow. This also seems to be relevant for chronically stressed humans and women in particular.
"Inappropriate" neurosteroids may be related to dysfunctional GABAergic transmission, and increased susceptibility to stress and developing psychiatric disorders. A recent paper states: "Altered expression of steroidogenic enzymes may warrant future detailed investigations."66 As we've seen, 5α-reductase is a critical, rate-limiting neurosteroidogenic en-zyme and it is worthy of attention.
Summary
Men and women have the same sex hormones, in differing amounts and relative ratios – vive la différence! Enzymes produce and process our hormones, controlling our physical form, and strongly influencing our moods, thoughts, and behavior. 5alpha-reductase is a critical enzyme that plays a role almost from conception. It strengthens the effects of testosterone and aldosterone and mitigates the strength of cortisol. It provides neurosteroids that alter the critical balance of neural stimulation and inhibition.
Oral testosterone replacement is ineffective because of the hepatic "first pass," which converts the testosterone to estrogens or DHT. It is now clear that the skin itself can have a first-pass effect. When transdermal testosterone replacement inappropriately induces 5α-R, consequences range from mild to drastic hormone imbalances and, when left uncorrected, can reduce life expectancy. This acquaintance with 5α-R provides an introduction to neurosteroids, a new frontier in psycho-neuroendocrinology. Men with robust 5α-R activity but limited progesterone substrate may suffer from the lack of 5α-reduced neurosteroids.
Segue to Women
The sequel to this article will review the importance of 5α-R and ALLO among women, whose problems can be both similar to and the opposite of men's. Consider an old puzzle: PMDD, PMS, and postpartum depression (PPD) are obviously associated with changing steroid sex hormone levels. However, all searches for a causal sex hormone abnormality yielded only a "frustrating lack of evidence."85 Through case presentations and literature review, it will be seen that investigating not just DHT but neurosteroids as "affective switches" is very rewarding. 5α-reductase blockers – even a simple herbal treatment – offer substantial relief for suffers of PMDD/PMS and dysmenorrhea.19
Notes .pdf
 Alan McDaniel, MD, is a 1977 Tulane University medical graduate. He trained in general surgery and emergency medicine before becoming board-certified in otolaryngology with subspecialties in neurotology and allergy. He has practiced privately since a two-year faculty appointment at the University of Louisville. He has presented at various national meetings in the US (AAO-HNS, AAOA, ANS, AAEM, IFM, PAAS) and Mexico. His topics have included otology and neurotology, allergy, chronic fatigue, and endocrinology. He has been a faculty member for the American Academy of Otolaryngic Allergy Basic and Advanced Courses and for the American Academy of Environmental Medicine. His two-day course "New Endocrinology" has been presented at the AAEM and elsewhere since 2005, to physicians from five continents. Work with dizziness and allergy in the 1980s led Dr. McDaniel to seek solutions for chronic fatigue syndrome. In turn, these investigations extended to the endocrine aspects of this and related conditions. Since basic surgical training emphasizes the need to know several alternative approaches to an operation, he saw the logic of studying integrative and controversial medical methods. He has endeavored to understand these in the light of new facts from research, mindful that medical history shows innovation begins as a minority opinion. He is excited that applying new research to patient care offers solutions to many of the chronic and worsening problems that are epidemic in modern society. Alan McDaniel, MD, is a 1977 Tulane University medical graduate. He trained in general surgery and emergency medicine before becoming board-certified in otolaryngology with subspecialties in neurotology and allergy. He has practiced privately since a two-year faculty appointment at the University of Louisville. He has presented at various national meetings in the US (AAO-HNS, AAOA, ANS, AAEM, IFM, PAAS) and Mexico. His topics have included otology and neurotology, allergy, chronic fatigue, and endocrinology. He has been a faculty member for the American Academy of Otolaryngic Allergy Basic and Advanced Courses and for the American Academy of Environmental Medicine. His two-day course "New Endocrinology" has been presented at the AAEM and elsewhere since 2005, to physicians from five continents. Work with dizziness and allergy in the 1980s led Dr. McDaniel to seek solutions for chronic fatigue syndrome. In turn, these investigations extended to the endocrine aspects of this and related conditions. Since basic surgical training emphasizes the need to know several alternative approaches to an operation, he saw the logic of studying integrative and controversial medical methods. He has endeavored to understand these in the light of new facts from research, mindful that medical history shows innovation begins as a minority opinion. He is excited that applying new research to patient care offers solutions to many of the chronic and worsening problems that are epidemic in modern society.
|
![]()
![]()
![]()






 Alan McDaniel, MD, is a 1977 Tulane University medical graduate. He trained in general surgery and emergency medicine before becoming board-certified in otolaryngology with subspecialties in neurotology and allergy. He has practiced privately since a two-year faculty appointment at the University of Louisville. He has presented at various national meetings in the US (AAO-HNS, AAOA, ANS, AAEM, IFM, PAAS) and Mexico. His topics have included otology and neurotology, allergy, chronic fatigue, and endocrinology. He has been a faculty member for the American Academy of Otolaryngic Allergy Basic and Advanced Courses and for the American Academy of Environmental Medicine. His two-day course "New Endocrinology" has been presented at the AAEM and elsewhere since 2005, to physicians from five continents. Work with dizziness and allergy in the 1980s led Dr. McDaniel to seek solutions for chronic fatigue syndrome. In turn, these investigations extended to the endocrine aspects of this and related conditions. Since basic surgical training emphasizes the need to know several alternative approaches to an operation, he saw the logic of studying integrative and controversial medical methods. He has endeavored to understand these in the light of new facts from research, mindful that medical history shows innovation begins as a minority opinion. He is excited that applying new research to patient care offers solutions to many of the chronic and worsening problems that are epidemic in modern society.
Alan McDaniel, MD, is a 1977 Tulane University medical graduate. He trained in general surgery and emergency medicine before becoming board-certified in otolaryngology with subspecialties in neurotology and allergy. He has practiced privately since a two-year faculty appointment at the University of Louisville. He has presented at various national meetings in the US (AAO-HNS, AAOA, ANS, AAEM, IFM, PAAS) and Mexico. His topics have included otology and neurotology, allergy, chronic fatigue, and endocrinology. He has been a faculty member for the American Academy of Otolaryngic Allergy Basic and Advanced Courses and for the American Academy of Environmental Medicine. His two-day course "New Endocrinology" has been presented at the AAEM and elsewhere since 2005, to physicians from five continents. Work with dizziness and allergy in the 1980s led Dr. McDaniel to seek solutions for chronic fatigue syndrome. In turn, these investigations extended to the endocrine aspects of this and related conditions. Since basic surgical training emphasizes the need to know several alternative approaches to an operation, he saw the logic of studying integrative and controversial medical methods. He has endeavored to understand these in the light of new facts from research, mindful that medical history shows innovation begins as a minority opinion. He is excited that applying new research to patient care offers solutions to many of the chronic and worsening problems that are epidemic in modern society.