Page 1, 2, 3
A research program called Prime investigated the relationship of several risk factors to IHD incidence and mortality rates in Northern Ireland and France.25 Data showed that cardiovascular event rates were 2.3 to 2.5 times more frequent in Belfast than in Lille or Strasbourg and 3.3 times more frequent in Belfast than in Toulouse.26 There were no important differences in macronutrient intake, although saturated fat intake was significantly higher in Belfast and dietary cholesterol was significantly higher in Toulouse. IHD mortality rate in Northern Ireland was about 3 times higher than in France without major differences in classical risk factors. The A1 beta-casein consumption in Northern Ireland is also about 3-fold higher than the cities in France (excluding cheese).
Around the world, there are also communities who are recorded as having extremely low rates of IHD but who drink milk, such as the Masai and Samburu in Kenya. They obtain their milk from Zebu cattle, which all contain the A2 beta-casein allele. Tibetan highlanders' source of milk, the yak, also does not have the beta-casein A1 allele and they share similarly low rates of IHD.27
With regard to pharmacological evidence, BCM-7 has also been shown to catalyze the oxidation of low density lipoprotein.28 Oxidized LDL (oxLDL) is considered a useful marker of heart disease, according to many cardiologists who consider it the main molecule responsible for initial injury to the cardiac endothelium.29 LDL oxidation is also considered to be one of the initiators of endothelial damage. In addition, oxLDL is associated with cholesterol accumulation in vascular walls, and elevated plasma oxLDL has been found in patients with arteriosclerosis.30,31 The tyrosine amino acid on the end of BCM-7 gives it strong oxidative capability, especially since the tyrosine radical has been found in atherosclerotic lesions.32 Torreilles and Guerin found that peptides from casein-derived peptides could act as a catalyst for the oxidation of human LDLs.33
A study in formula-fed human infants found elevated serum levels of antibodies to oxLDL relative to breast-fed babies.34 The formula contained A1 milk. Since BCM-7 has already been shown (above) to oxidize LDL, it is possible that the BCM-7 present in the formula caused the oxidation of the LDL molecules. Although the researchers in this study did not assess serum BCM-7 levels in these infants, they suggested, "As human milk does not contain beta-casein A1 and infant formulas are based on bovine milk, we can express a hypothesis that beta-casein A1 is the substance, which caused increased production of IgoxLDL."35
Annand has pointed out that the introduction of pasteurization in the UK around the early 1920s coincided with a near doubling of heart disease mortality.36 Holder pasteurization, which caused milk to develop a slightly cooked flavor, was generally replaced by the high-temperature short-time (HTST) pasteurization process in the late 1940s to mid 1960s, depending on the country. HTST pasteurization was almost universally adopted by 1980.37 The US was the first country to introduce pasteurization of milk, around 1900. Ostler in 1910 reported an increased heart disease prevalence that had been observed in the US.38 It is unknown which of these two possible effects of HTST, whether the heat treatment of milk prior to consumption affects the transmission of b-casein through the gut wall or whether it contributes to greater production of specific casein fragments, is the main culprit and this remains to be established. However, the two factors, changes in pasteurization and differing b-casein allele frequencies between areas, in conjunction with changes in traditional risk factors, may provide an explanation for the historical changes in IHD and regional variations in the disease.
Type 1 Diabetes
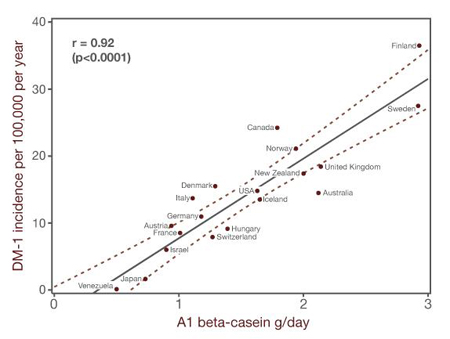
Ecological evidence across 20 developed nations show strong correlations between the consumption of A1 beta-casein and the incidence of DM-1.39–42 Figure 6 shows this nicely.43 In addition, a recent study reported that A1 beta-casein consumption during early childhood may be more important than during adolescence for DM-1 development. Birgisdottir et al. (2006) compared A1 beta-casein consumption among 2-year-olds and among 11- to 14-year olds in Iceland and Scandinavia (i.e., Norway, Denmark, Sweden, and Finland) and evaluated this against the incidence of DM-1.44 For the 2-year-olds, but not the 11- to 14-year olds, A1 beta-casein consumption correlated strongly with DM-1 incidence (r = 0.9; p = 0.037). They raise the possibility that intensive dairy cattle breeding may have emphasized a genetic variant in milk with adverse effects in humans. Further animal research and clinical trials would be needed to compare disease risks of A1-free versus "ordinary" milk.
Figure 6
A limited number of human trials also suggest that beta-casein may stimulate a T-cell immune response or an antibody immune response in the development of DM-1.45–48 For instance, Monetini et al. (2001) showed significantly higher levels of antibodies to beta-casein in bottle-fed infants under 4 months of age compared with exclusively breast-fed infants (p < 000.1) and significantly higher levels of antibodies in prepubertal children with DM-1 compared with age-matched controls (p = 0.03).49 How this should be interpreted is open to debate. It is possible that this is a manifestation of those with DM-1 being particularly sensitive to antibody reactions. However, in one of the only human studies to investigate differences in antibody response to A1 and A2 beta-casein, Padberg et al. (1999) showed that the ratio of A1 to A2 beta-casein antibodies was higher in those with DM-1 compared with case controls (p < 0.001).50 These results suggest that A1 beta-casein may be a modifier in the development of DM-1 in "at risk" individuals.
One mechanism by which A1 beta-casein may contribute to the development of DM-1 relates to the potential molecular mimicry (or cross-reactivity) between beta-casein and an epitope of the pancreatic beta-cell glucose transporter GLUT2, as autoantibodies to GLUT2 have been described in patients with recent-onset DM-1.51–53
A second mechanism involves the potential antigenic determination characteristic of beta-casein, which may lead to the autoimmune destruction of pancreatic beta-cells.54 More specifically, Cavallo et al. (1996) have suggested that there may be molecular mimicry between a sequence of the beta-casein protein and an epitope of the GLUT2 transporter, which may give rise to autoantibodies capable of targeting pancreatic beta-cells.55
Currently, the role of bovine BCM-7 in the health and development of human infants is the topic of extensive scientific debate. Such debate was stimulated recently with the publication by Russian scientists, Kost et al. (2009), who found that BCM-7 could be measured in the blood of infants fed cow's milk formula.56 The higher blood levels of bovine BCM-7 found in some infants correlated with delays in psychomotor development.57
Clinical Stories
I have personally seen some people (not all!) who switched from A1 to A2 milk and had their milk intolerance disappear. One patient was a Czech who had escaped in 1962 from Czechoslovakia, was raised in Switzerland, and came to the US as an adult. He could not tolerate any dairy products here. He went to a farmers' market in Portland, Oregon, where he saw advertised that they only sold milk from A2 cows. He decided to try their cheese and milk and, voilà, his milk intolerance was gone, as long as he only drank the A2 milk.
Another patient has found that his severe chronic lower back and morning stiffness has almost completely disappeared after he switched to only A2 milk. These symptoms had been present almost continuously for 20 years. He feels as if a miracle has happened.
Another patient said she used to avoid drinking milk in the evenings because it would make her legs jerky. She has now consumed A2 milk in the evening several times and that has not happened. On a recent night she had symptoms again and thought that maybe it was not the A1 after all. Then she remembered that she had eaten a salad with feta cheese that was made from A1 milk.
Potential Genetic Solution
The solution to the problem is both simple and unbelievably cheap. All that is required is for farmers to ensure that their cows are inseminated, naturally or artificially, with semen from A2/A2 bulls. In New Zealand, some of the smaller groups of dairy farmers, predicting the increase in consumer demand for A2 milk, have already converted their herds to A2 cows.
The A1 gene can be bred out of a herd in about 10 to 15 years simply by choosing what are called A2/A2 sires. This means that neither the dam or sire carries the A1 gene.58 Unfortunately, until the consumer is educated and begins to request A2 milk, the motive will not be there for the selective breeding in most dairies. Interestingly, New Zealand labels A2 milk in its grocery stores.
What can you do? You can ask your providers of local milk if they are breeding for A2/A2 milk. If they do not understand it, you can refer them to this article. You could also consider purchasing your own milk goats (they do not carry the A1 gene), buy a cow with A2 genetics, or buy a cow with the plan to breed A2 genetics and select future heifers.
Page 1, 2, 3 |
![]()
![]()
![]()





