Page 1, 2
Pattern V
Pattern V displays a flattened insulin curve, with all insulin values being less than 30 (Figure 9). This is considered to be an inadequate insulin response to the glucose challenge and suggests exhaustion of pancreatic islet cells. This might be seen in someone who has been hyperinsulinemic for an extended period of time and now has a decreased capacity to respond. Typically, glucose values will be in diabetic ranges if not otherwise controlled.
In a few cases, Pattern V insulin response will be seen in conjunction with normal glucose levels. This may be due to a low-carbohydrate diet that has resulted in a downregulated insulin response.
Figure 9
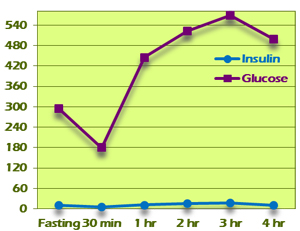 |
Figure 10
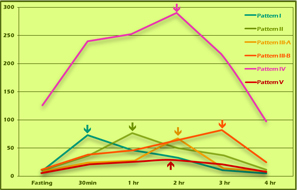 |
GTIR Pattern Progression
Putting the insulin curves for the different patterns into a single graph illustrates a distinct progression of insulin resistance from normal to insulinopenic (Figure 10). With this test, nascent insulin resistance can be detected long before blood glucose values might start to sound alarm bells. The import of this is magnified when one considers that diabetes has both individual and societal costs, and that it can largely be prevented or reversed with earlier detection, lifestyle changes, and treatment.
Case Study
The value of the GTIR for early detection and treatment cannot be overstated. The case of G. J. is a compelling example of this. G. J. is a 38-year-old woman who came into our clinic with a chief complaint of easy weight gain and fatigue. She is 5'4" tall and weighed 174 pounds at the initial visit. BMI was 29.9. Her pulse and respirations were normal and her BP was 107/76. She had a history of gestational diabetes and a family history of type 2 diabetes. Fasting blood sugar was elevated at 111, but HgbA1c was only 5.3. Because of the family and personal history, a 4-hour GTIR test was run. On the test, fasting and 2-hour glucose were 83 and 113, respectively, both well within the limits of normal based on American Diabetes Association criteria. Fasting insulin was above 10 (11.80) and peaked in the second hour at 93.20. This is a Pattern IV insulin response (Figure 11A).
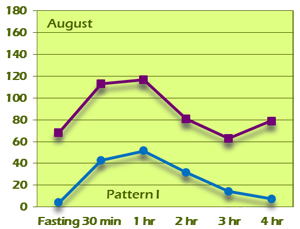
G.J. was put on berberine, 500mg t.i.d., and counseled about diet and exercise. She was highly motivated because of her Pattern IV GTIR result. At her 6-month follow-up visit, she had lost 9 pounds and her BMI had decreased to 28.3. Her HgbA1c was also improved at 5.1. Her GTIR test demonstrated a dramatic reversal from the original Pattern IV result to a completely normal Pattern I result (Figure 11B).
Figure 11A
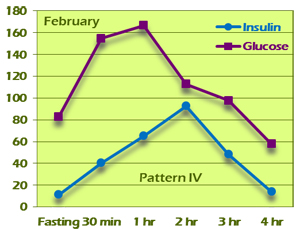 |
Figure 11B
 |
A New Method of GTIR Testing
Up until now, the GTIR test has required the capacity to do multiple venipunctures over an extended period of time, whether in the practitioner's office or at a lab draw station. Now a new finger-stick version of the test is being introduced, making early detection of insulin resistance accessible to those practitioners who do not draw blood in their offices. Finger-stick blood sugar measurements have been around for decades, of course, and finger-stick testing of insulin is not new. However, inherent differences between venous blood and capillary blood in both sugar and insulin levels require careful calibration of reference ranges to allow accurate identification of the Kraft patterns of insulin response. The new blood-spot GTIR test is the result of extended testing and verification to authenticate these patterns.
The Cost of Ignorance
It is indisputable that rising costs have the US health-care system teetering on the edge of catastrophe. It can certainly be argued that this is in large part because of the focus on "disease management" rather than actual "health care" or prevention. With any condition, early detection allows for early intervention. The earlier the intervention, the fewer drastic measures are needed and the better the chances for a return to health. In the US, 21 million adults have been diagnosed with diabetes and another 8.1 million have diabetes and are undiagnosed. In addition, it is estimated that 86 million Americans over age 20 have prediabetes, based on fasting glucose or HgbA1c levels.5 Yet as we have seen in the case of G. J. and other examples shown in the graphs presented, these parameters miss people who show signs of growing glucose/insulin dysregulation if the insulin response is taken into account. What is the price of this ignorance?
The total estimated cost of diabetes in the US in 2012 was $245 billion. That figure includes direct medical costs as well as indirect costs such as disability and loss of income due to missed work.5 If we extend those costs to the 86 million with prediabetes, we are looking at more than $700 billion in additional future costs. This does not include those with insulin resistance who slip under the ADA radar.
After adjusting for age and sex differences, the average medical expenses among people diagnosed with diabetes was 2.3 times higher than those without diabetes.5 If we could prevent only those 86 million with prediabetes from progressing to diabetes (and perhaps even reverse their condition), that would translate into nearly half a billion additional dollars that these individuals could use in more productive ways. These numbers also do not account for the more human costs, in decreased function, enjoyment of life, and ability to contribute to one's community, that accompany chronic disease.
Current thinking about reducing health-care costs in the US focuses on reducing testing (deemed "unnecessary" testing). There is evidence that this may also be the case in Canada. This is a penny-wise, pound-foolish approach. It saves money now, but at the cost of billions of dollars of future health-care expenses. To truly reduce health-care costs requires preventing chronic diseases from developing in the first place. Diabetes is one such disease wherein the natural progression of the disease is clear enough to make early detection too valuable a tool to omit.
Who Should Be Tested?
The National Diabetes Education Program recommends that anyone with risk factors be evaluated for diabetes. Besides the obvious risk factors, such as family history, gestational diabetes, lipid abnormalities, or elevated blood pressure, the NDEP also recommends including African American, Hispanic/Latino, American Indian, Asian American, or Pacific Islander ethnicity as triggers for increased vigilance. A BMI of >25 (>23 for Asian, >26 for Pacific Islander) is also a reason for further evaluation. Simply being 45 years or older warrants increased surveillance. Also on the NDEP risk factor list are PCOS, acanthosis nigricans, history of giving birth to a baby of 9 pounds or more, and being physically active less than 3 times a week.6
Not on the NDEP list but worthy of consideration in this context are tinnitus, sugar cravings, symptoms of hypoglycemia, sleep disturbances (including in those who are shift workers and others with disrupted sleep patterns), skin tags, osteoarthritis prior to age 50, Peyronie's disease, DuPuytren's contracture, recurrent yeast/fungal infections, changes in vision, gum disease, and low testosterone in men. All of these conditions or symptoms have been associated with changes in blood sugar and insulin regulation.7–19
It is also worth noting that patients with an optimal BMI may still exhibit central weight gain. Thin patients who also have "love handles" or a "muffin top" may be showing signs of insulin resistance (which may be well disguised by their clothing). Certainly any weight gain beyond normal growth in a child or adolescent should raise red flags.
Beyond the more familiar laboratory markers discussed earlier, a number of other less than optimal results point to insulin resistance. In a 24-hour urine hormone profile, elevated 5a-reductase, a testosterone:estrogen ratio of <4 (in men), an elevated cortisol/cortisone ratio, and elevated cortisol metabolites all suggest further evaluation for insulin resistance. Hyperviscosity on a blood viscosity panel may also have insulin resistance as an underlying etiology.
For an individual with a healthful lifestyle and no other risk factors, a baseline GTIR at 45 years of age would be prudent, with follow-up testing every 3 to 5 years if no signs of insulin resistance become apparent.
There is no question that the problem of type 2 diabetes has reached epidemic proportions. This disease takes a great toll, both personal and societal, and reducing the incidence of diabetes would provide wide-ranging benefits. The best way to do this is by preventing the development of the disease in its earliest stages, long before it actually becomes diabetes. Traditional methods of detection are good but miss many people in the early stages of insulin resistance. The glucose tolerance/insulin response test offers a way to improve our ability to intervene earlier, when it can make the most difference.

Notes
1. About diabetes [Web page]. World Health Organization. http://www.who.int/diabetes/action_online/basics/en. Oct. 6, 2014.
2. American diabetes association: diagnosing diabetes and learning about pre-diabetes [Web page]. Sept. 22, 2104. www.diabetes.org/diabetes-basics/diagnosis/?loc=db-slabnav Accessed Oct. 6, 2014.
3. Kraft JR. Detection of diabetes mellitus in situ (occult diabetes). Lab Med. 1975;6(2):10–22.
4. Kraft JR. Diabetes Epidemic and You. Bloomington, IN: Trafford; 2009.
5. National diabetes statistical report, 2014 [online document]. CDC. https://www.cdc.gov/diabetes/pdfs/data/2014-report-estimates-of-diabetes-and-its-burden-in-the-united-states.pdf. Accessed May 12, 2018.
6. Diabetes risk factors [Web page]. National Diabetes Education Program. http://ndep.nih.gov/am-i-at-risk/DiabetesRiskFactors.aspx. Accessed Oct. 1, 2014.
7. Kraft JR. Hyperinsulinemia: a merging history with idiopathic tinnitus, vertigo, and hearing loss. Int Tinnitus J. 1998;4(2):127–130.
8. Lavinsky L et al. Hyperinsulinemia and tinnitus: a historical cohort. Int Tinnitus J. 2004;10(1):24–30
9. Spiegel K et al. Sleep loss: a novel risk factor for insulin resistance and Type 2 diabetes. J Appl Physiol. 2005;99(5):2008–2019.
10. Buxton O et al. Sleep restriction for 1 week reduces insulin sensitivity in healthy men. Diabetes. 2010;59(9):2126–2133.
11. Spiegel K et al. Impact of sleep debt on metabolic and endocrine function. Lancet. 1999;354(9188):1435–1439.
12. Schilling W, Crook M .Cutaneous stigmata associated with insulin resistance and increased cardiovascular risk. Int J Dermatol. 2014;53(9):1062–1069.
13. Sellam J, Berenbaum F. Is osteoarthritis a metabolic disease? Joint Bone Spine. 2013;80(6):568–573.
14. Han CD et al. Correlation between metabolic syndrome and knee osteoarthritis: data from the Korean National Health and Nutrition Examination Survey (KNHANES). BMC Pub Health. 2013;13:603.
15. Deveci S et al. Defining the clinical characteristics of Peyronie's disease in young men. J Sex Med. 2007;4(2):485–490.
16. Papanas N et al. The diabetic hand: a forgotten complication? J Diabetes Complications. 2010;24(3):154–162.
17. Kolar P. Risk factors for central and branch retinal veign occlusion: a meta-analysis of published clinical data. J Ophthalmology. 2014.
18. Demmer R et al. Periodontal infection, systemic inflammation, and insulin resistance: results from the continuous National Health and Nutrition Examination Survey (NHANES) 1999–2004. Diabetes Care. 2012;35(11):2235–2242.
19. Kapoor D et al. Androgens insulin resistance and vascular disease in men. Clin Endocrinol. 2005;63(3):239–250.
 Dr. Pushpa Larsen graduated from Bastyr University with a doctorate in naturopathic medicine and certificates in naturopathic midwifery and spirituality, health and medicine. She has worked as a research clinician for the Bastyr University Research Institute and as affiliate clinical faculty for Bastyr University, training students in her clinic. She was in private practice in Seattle for 10 years before joining Meridian Valley Lab as a consulting physician, and maintains a small private practice primarily focused on bioidentical hormone therapy. She consults with hundreds of doctors every year on the use and interpretation of 24-hour urine hormone profiles, as well as other tests offered by Meridian Valley Lab. She has previously published and presented on the use of carotid intima-media thickness (CIMT) scanning in primary-care settings for early detection of cardiovascular disease. She can be reached at plarsen@meridianvalleylab.com or at 425-271-8689. Dr. Pushpa Larsen graduated from Bastyr University with a doctorate in naturopathic medicine and certificates in naturopathic midwifery and spirituality, health and medicine. She has worked as a research clinician for the Bastyr University Research Institute and as affiliate clinical faculty for Bastyr University, training students in her clinic. She was in private practice in Seattle for 10 years before joining Meridian Valley Lab as a consulting physician, and maintains a small private practice primarily focused on bioidentical hormone therapy. She consults with hundreds of doctors every year on the use and interpretation of 24-hour urine hormone profiles, as well as other tests offered by Meridian Valley Lab. She has previously published and presented on the use of carotid intima-media thickness (CIMT) scanning in primary-care settings for early detection of cardiovascular disease. She can be reached at plarsen@meridianvalleylab.com or at 425-271-8689.
Page 1, 2 |
![]()
![]()
![]()









 Dr. Pushpa Larsen graduated from Bastyr University with a doctorate in naturopathic medicine and certificates in naturopathic midwifery and spirituality, health and medicine. She has worked as a research clinician for the Bastyr University Research Institute and as affiliate clinical faculty for Bastyr University, training students in her clinic. She was in private practice in Seattle for 10 years before joining Meridian Valley Lab as a consulting physician, and maintains a small private practice primarily focused on bioidentical hormone therapy. She consults with hundreds of doctors every year on the use and interpretation of 24-hour urine hormone profiles, as well as other tests offered by Meridian Valley Lab. She has previously published and presented on the use of carotid intima-media thickness (CIMT) scanning in primary-care settings for early detection of cardiovascular disease. She can be reached at
Dr. Pushpa Larsen graduated from Bastyr University with a doctorate in naturopathic medicine and certificates in naturopathic midwifery and spirituality, health and medicine. She has worked as a research clinician for the Bastyr University Research Institute and as affiliate clinical faculty for Bastyr University, training students in her clinic. She was in private practice in Seattle for 10 years before joining Meridian Valley Lab as a consulting physician, and maintains a small private practice primarily focused on bioidentical hormone therapy. She consults with hundreds of doctors every year on the use and interpretation of 24-hour urine hormone profiles, as well as other tests offered by Meridian Valley Lab. She has previously published and presented on the use of carotid intima-media thickness (CIMT) scanning in primary-care settings for early detection of cardiovascular disease. She can be reached at