By Thierry Hertoghe, MD
Cortisol is essential for human life. To stay alive, humans need cortisol. Life is not possible without it. In the absence of cortisol, the sugar level drops below 30 mg/dl, and the systolic blood pressure drops below 50 mmHg, which will cause a patient to fall into a deep coma and die about 24 hours later(1-3).
Cortisol is also essential to enjoy a good quality of life(4-7). When the cortisol level drops below a threshold in the blood, life is experienced as increasingly more difficult and stressful. If the level of cortisol drops further, life literally becomes miserable and plagued with anxiety, fatigue, and suffering.
Frequency of Cortisol Deficiency in an Ambulatory Setting
How many individuals in a population are cortisol-deficient? Data about the frequency of cortisol deficiency is scarce, if nonexistent. One study reported that 7% of hospitalized patients received glucocorticoid therapy during their stay(8), but this percentage probably does not reflect the incidence of permanent states of cortisol deficiency in the global population.
An epidemiological study covering the whole population of Iceland over 18 years of age concluded that only 0.02% (22.1 per 100,000) of the population were suffering from primary adrenal deficiency(9). Spanish researchers found an even lower prevalence of 0.01% (10 cases per 100,000) in Spain suffering from Addison’s disease(10). These low numbers are based on the extremely low number of patients receiving the diagnosis of Addison’s disease and treatment for it in Iceland and Spain. Most cases are likely diagnosed with the traditional ACTH test, which is done with an inaccurate 250-fold too high dose (read further for more information). The numbers probably indicate that local physicians are not accustomed in diagnosing and treating primary adrenal insufficiency, leaving many cases of Addison’s disease undiagnosed.
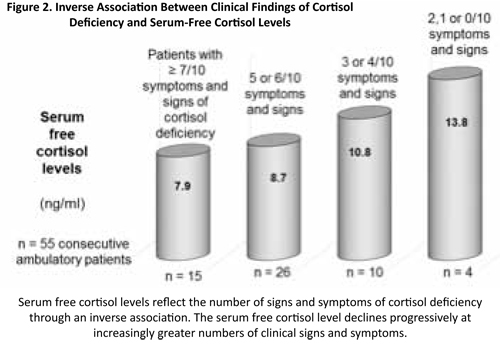
To get more accurate information through broader investigations including both clinical and laboratory findings, together with my colleagues of the clinic, we investigated the prevalence of cortisol deficiency in 56 consecutive and untreated (not receiving any glucocorticoid treatment) patients visiting our clinic for hormone therapy for the first time (36 females, mean age: 50.6 years (20-78); 20 males, mean age: 42.3 years (20-61)). Depending on the severity of clinical and laboratory diagnostic criteria, 4%, 13%, 50%, or 84% of these patients can be considered cortisol-deficient and could potentially benefit from cortisol treatment(11).
Diagnosis was based on an association of both clinical findings and laboratory test results, and not on a single laboratory test or sets of signs and symptoms alone without a suggestive laboratory test.
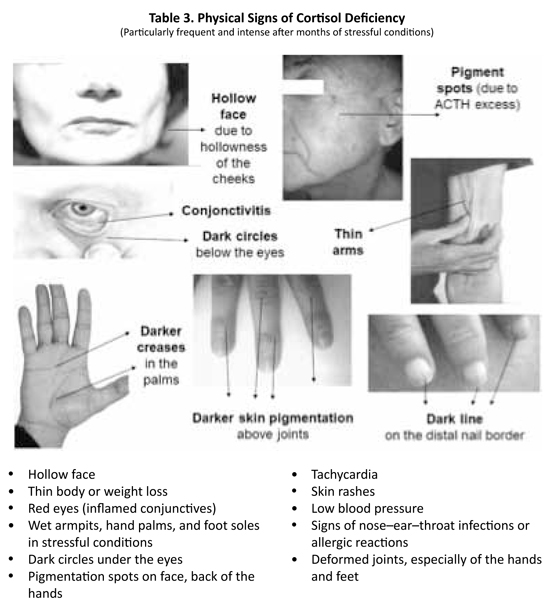
The following five symptoms of cortisol deficiency were checked through a questionnaire filled in by the patient: low resistance to stress, fatigue in stressful situations, low blood pressure, sugar cravings, and allergic reactions (skin, ENT, and/or asthma). The five physical signs of cortisol deficiency that were assessed were hollow face, pigmented spots on the face, conjunctivitis, dark circles under the eyes, and wet palms (due to peak secretions of adrenaline and noradrenaline in stress situations compensating for the low cortisol levels). Thus, a list was made of five symptoms and five signs suggestive of cortisol deficiency. The simple presence of one of the complaints or signs, whatever its intensity, was considered as contributing to (but not establishing) the diagnosis of cortisol deficiency.![]()
The laboratory tests checking for diagnosis of cortisol deficiency included serum-free cortisol, 24-hour urinary-free cortisol, and 24-hour urinary 17-hydroxysteroids (i.e., the cortisol metabolites). We considered any test value below a critical level in laboratory tests as suggestive of (but not decisive for) the diagnosis of cortisol deficiency. The critical level was a level halfway between the average and the lower reference limit of the laboratory. The reason we used a critical level that is above the lower reference limit and not the lower limit itself is that the lower reference limit is a statistical – and not a health limit – and that many lower levels within the reference range are significantly associated with disease. These relationships are explained further in the paragraph on “Cortisol Reference Ranges.” For serum-free cortisol, the cutoff level was 12 (reference range: 7-27 ng/mL). For 24-hour urinary-free cortisol, the threshold was 30 (reference range: 10-90 µg/24 h). For the 17-hydroxycorticoids in 24-hour urines, the limit was fixed at 4.54 (reference range: 3.17-8.63 mg/24 h) in females and 7.5 (reference range: 6.10-11.70 g/24 h) in males.
Four categories of diagnostic criteria of increasing severity were applied to establish the prevalence. All of these criteria categories have medical legitimacy for diagnosis of cortisol deficiency.
The first type of diagnostic criteria is the presence of at least three of the ten possible signs or symptoms, with at least one of the three laboratory test results suggestive of cortisol deficiency. As shown in Figure 1, 84% of our patients fit into this category.
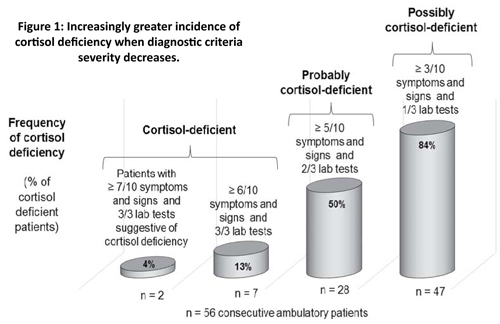
The second type necessitated the presence of at least five of the ten possible signs or symptoms, with at least two of the three laboratory test results suggestive of cortisol deficiency. As shown in Figure 1, half of the patients belonged to this category.
Criteria of the third category were severe. Only 13% of the patients presented the required minimum of six of the ten possible signs or symptoms and all three laboratory tests suggestive of cortisol deficiency, and could be classified in this group of undoubtedly cortisol-deficient patients. Only 4% (2 of 56) of patients belonged to the fourth group of severe diagnostic criteria with seven or more symptoms and signs and all three laboratory tests being conclusive.
The results of this small study show a high frequency of cortisol deficiency, which increases when criteria are less severe. They should not be considered representative of the general population but merely illustrative of the incidence of adrenal deficiency in a selected group within the general population, i.e., those individuals who are sent or go spontaneously to a clinic focusing on hormone therapy. The frequency of cortisol deficiency within this group is almost certainly greater than in the general population because many of our patients had levels in the lower third of the reference range in the laboratory tests(11).
Incidence of Cortisol Deficiency with Aging
Some studies have shown lower serum cortisol levels with aging(12-13), others no change(14), while several investigations showed higher levels, especially in the evening(15-16), which is at a time when the cortisol levels should considerably drop to allow the body to calm down and relax and prepare for sleep. However, as the metabolic clearance of cortisol decreases with age, thus allowing cortisol to remain for a longer time in the blood and less in the target cells(17-19), the higher evening cortisol level may not reflect a higher cortisol activity in the target cells.
On the contrary, cortisol metabolic activity may substantially decrease with age following a study that showed that the 24-hour urinary excretion of the 17-hydroxycorticoids – the main metabolites of cortisol (and, thus, a reflection of cortisol metabolic activity in target cells) – are nearly 80% lower: 2 mg/day in older adults compared to 9.3 mg in young adults!(20)
The Great Cortisol Discovery
In the mid-1930s, Kendall and Reichstein isolated several hormones of the adrenal glands, including compound E or cortisol, which was the most effective in prolonging the life of adrenalectomized animals. In 1946, Louis Sarettt succeeded in synthetizing cortisol for the first time. In 1948, sufficient quantities of compound E became available for the rheumatologist Philip Hench to test it successfully for the first time in a patient with rheumatoid arthritis(21-23). The improvement in this patient and others was so impressive that it triggered enthusiasm all over the medical world so that, in 1950, Kendall, Reichstein, and Hench soon after received the Nobel Prize in medicine for their work on cortisol.
However, three errors were and still are made with cortisol treatment, which have triggered controversies on cortisol’s use ever since and have probably made cortisol the most misunderstood and unjustly rejected treatment in medicine.
Three Therapy Errors That Spoil Outcome
What are these common errors? Firstly, excessive doses of cortisol were administered to quickly treat rheumatoid and other inflammatory disorders. Prolonged use of high cortisol doses reduces skin thickness, muscle volume, and bone density and provokes swelling, weight gain, bruising, pathological effects on the cardiovascular system, and at times a euphoric, excessively agitated character(8,24-27) in contrast, low doses of cortisol (i.e. 20-30 mg/day of hydrocortisone) or of one of its derivatives (5 mg of prednisolone for example) are relatively safe(28-31). In septic shock, a low dose appears to show efficacy in decreasing mortality, whereas a high dose does not(32-33).
Secondly, cortisol or its glucocorticoid derivatives were administered alone without the protection of other hormone supplements, such as DHEA (dehydroepiandrosterone – the other primary adrenal hormone), that prevents any excessive tissue catabolism from cortisol. In healthy individuals, each time the adrenal glands secrete the catabolic hormone cortisol, they also secrete protective anabolic hormones such as DHEA, androstenedione, and other androgens(19,34-39).