Page 1, 2
Masking
Suppose that TILT was a mechanism underlying certain cases of chronic fatigue,
migraine, asthma, or depression. It might be reasonable to wonder, then,
why patients experiencing these symptoms do not also report chemical intolerances.
In fact, some but not all patients do report them.21,22 Many chemically sensitive
patients with these same diagnoses report that it was not until they accidentally
or intentionally avoided a sufficient number of their problem incitants that
they noticed feeling better. Then, when they reencountered one of those incitants,
robust symptoms occurred. As they repeated this iterative process of avoidance
and reexposure, they noticed that particular symptoms occurred with particular
exposures. Most indicate that had they not avoided many chemicals and foods
simultaneously, or unmasked themselves, they might not have determined what
was making them sick.
Masking and unmasking are colorful lay terms for which there is no scientific
equivalent. Nevertheless, investigators' abilities to understand masking
and unmasking and manipulate these variables knowledgeably may determine the
success of studies in this area. When chemically sensitive patients follow
a diet free of their problem foods and live in a relatively chemical-free home
in the hills of central Texas where there are no major agricultural or industrial
operations or air contaminants, they say they are in an unmasked state. Under
these circumstances they claim that if a diesel truck drove by they could identify
specific symptoms due to the diesel exhaust, for example, irritability, headache,
or nausea.
On the other hand, the patients report that when they travel to a large city
like Houston or New York City, stay in a hotel room, and eat in restaurants,
they become masked. In the presence of many concurrent exposures (exhaust,
fragrances, volatiles offgassing from building interiors, various foods) in
New York City, many report feeling chronically ill, as if they had flu. If
a diesel truck drove by under these circumstances, most report they would not
be able to attribute any particular symptoms to the exhaust because of background
noise from overlapping symptoms occurring as a consequence of overlapping or
successive exposures. In theory, such background noise, or masking, hides the
effects of individual exposures – responses are blurred.
Masking appears to involve at least three interrelated components, any of which
may interfere with the outcome of low-level chemical challenges in these individuals:
acclimatization, apposition, and addiction. In real life, these three components
probably operate concurrently, although here they are considered individually.
There is some notation that can be used to help depict these components. In
the addiction literature, responses to addictive drugs are often illustrated
graphically using a biphasic curve or sine wave (Figure 3). The portion of
the sine wave above the horizontal axis represents symptoms with onset of exposure,
often called stimulatory symptoms; the portion below the horizontal axis represents
symptoms with offset or cessation of exposure, often referred to as withdrawal
symptoms. The height or amplitude of the sine wave in either direction is proportional
to the severity of the response. For persons not sensitive to a particular
substance, the curve would be a flat line with zero amplitude in either direction.
The length of the biphasic curve represents the duration of symptoms following
an exposure, reportedly ranging from minutes up to several days depending upon
the exposure and the individual. Of course, the particular nature of the symptoms
vary from one sensitive subject to the next and from substance to substance.
Figure 3. Graphic
representation of symptom progression following exposure to a single substance
in a person sensitive to that substance
(e.g., caffeine, a solvent, alcohol, nicotine).
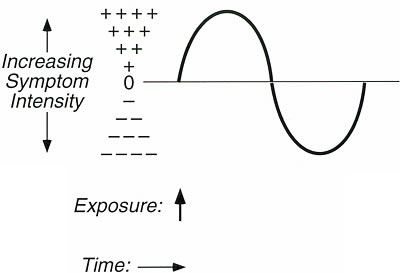
The portion of the biphasic
curve above the line represents symptoms with onset of exposure (stimulatory
symptoms) and the portion of the curve below the line represents symptoms with
offset of exposure (withdrawal symptoms). Amplitude is proportional to symptom
severity. The length of the curve (duration of symptoms) may range from minutes
to days.
Suppose researchers wished to test a putatively sensitive subject by exposing
him to a low concentration of xylene. Xylene is a common indoor air contaminant
and a component of Molhave's mixture23 that has been used in human inhalation
challenge studies. How would the researchers ensure that their subject was
unmasked (at true baseline) before challenge? The following components of
masking would need to be considered and controlled:
Acclimatization. For most of the population, with continuous or repeated
exposure to many environmental stressors, adaptation occurs. That is, symptoms
diminish
as exposure continues. Chemically sensitive patients' symptoms also decrease
with continuing exposure; however, when exposure ceases, these individuals
often report marked withdrawal symptoms. Thus, what they describe is more
akin to habituation than to adaptation. Suppose further that the subject
who is
challenged with xylene works in a sick building where he is routinely exposed
to low levels of xylene on a regular basis. Administering a test exposure
of xylene below the odor threshold (0.62 ppm)24 may produce little or no
effect
on the subject if he has been working in that same building during the preceding
week (Figure 4). On the other hand, if he avoided the building and all other
sources of xylene for four to seven days before testing, a more robust response
to the xylene challenge might be anticipated.
Thus, a sensitive subject's response to a challenge may range widely
in intensity, from none to maximal, depending on how recently that person
has been exposed to the test substance or a chemically related substance.
If insufficient
time has elapsed – for example, less than four days – the challenge
may yield a false negative response as a result of habituation. If too much
time has elapsed – for example, weeks or months – sensitivity
may have waned.
Figure 4. Graphic representation of acclimatization.
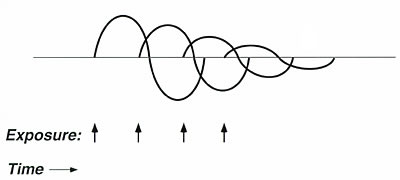
Symptom severity decreases
with repeated closely timed exposures (inhalant or ingestant) to the same
substance. Acclimatization is not equivalent to adaptation,
since patients report withdrawal symptoms after exposures cease; conceptually,
acclimatization more closely resembles habituation in this case.
Apposition. Suppose next that the research
subject is sensitive to multiple substances. On the day he is scheduled
for challenge testing, he gets up
in the morning, uses some scented soap or hair spray, cooks breakfast
on a gas
stove, and drives his car through heavy traffic to reach the laboratory.
Inside the laboratory building he rides an elevator where he is exposed
to people
wearing various colognes. If he were sensitive to several of these exposures,
his responses might overlap in time. Such responses reportedly can last
for hours or days. If this is true, they could persist during a placebo
challenge,
resulting in a false-positive response. Thus, apposition or juxtaposition
of the effects of closely timed exposures is a second component of masking
that
must be controlled prior to and during challenge studies (Figure 5).
Figure 5. Graphic representation of apposition.
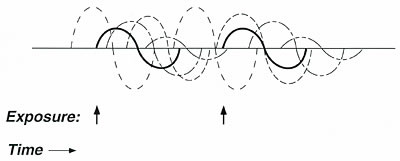
If an individual is sensitive
to many different substances, the effects of everyday exposures to chemicals,
foods, or drugs may overlap in time. This
apposition of effects might lead to an individual who feels ill most of the
time; however, neither the individual nor his physician notices the effect
of any single exposure. Apposition tends to mask the effect of interest (solid
lines) in much the same way that background noise masks a sound of interest.
Addiction. Many of the symptoms
reported by chemically sensitive patients mirror those commonly
associated
with addiction. Addiction may be a component
of masking.
Addicted individuals consciously or subconsciously time their
next "hit" so
as to forestall withdrawal symptoms (Figure 6), a phenomenon
that occurs in alcohol, tobacco, and caffeine addictions. However,
addiction
to
foods also
is reported among chemically sensitive patients. Randolph described
wheat, eggs, milk, and corn as the most common addictants in
his patients.14,17 Frequently, these individuals report intense
cravings
and consume astounding
quantities
of foods, for example, a pound of chocolate, several bags of
popcorn, a
dozen doughnuts, or 30 cups of coffee in one day. Patients
most often report this
kind of addictive consumption in the early stages of their
illness, before they practiced avoiding problem exposures.
Foods may contain bioactive constituents such as tyramine, monosodium
glutamate, and opiates.13 Persons who routinely use tobacco, caffeine,
alcohol, or foods
containing bioactive substances may need to avoid these substances
before testing because the pharmacologic effects of these agents
could override
or mask the
effect of an experimental challenge. Failure to eliminate addictants
before testing could result either in false-positive challenges,
due to lingering
symptoms from an addictant used in the hours or days preceding
a placebo challenge, or in false-negative challenges due to masking
by an addictant.
Figure 6. Graphic representation of addiction.
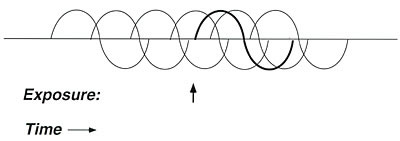
A
sensitive person who is addicted to caffeine, alcohol, nicotine, or another
substance may deliberately take that substance at frequent, carefully spaced
intervals to avoid unpleasant withdrawal symptoms. Such exposures may also
mask the effect of interest (e.g., a challenge test using xylene).
Testing the TILT Theory
After the germ theory of disease was introduced in the late 1800s, many overly
enthusiastic investigators who were careless in their bacteriological techniques
announced they had discovered causative agents for tuberculosis, yellow fever,
and other diseases. These pronouncements and subsequent retractions became
so frequent that in 1884 the President of the New York Academy of Medicine
lamented that a bacteriomania had swept over the medical profession.25 To
prevent future such pseudodiscoveries, Robert Koch, who identified the organisms
responsible for tuberculosis, anthrax, and cholera, proposed a set of rules
for etiological verification. His postulates required the following: the
microbe must present in every case of the disease; it must be isolatable
in pure culture; inoculating a healthy animal with the culture must reproduce
the disease; and the microbe must be recoverable from the inoculated animal
and be able to be grown again.
Just as bacteriomania engulfed the medical profession in the 1880s, chemomania
is poised to engulf it now. Chemical sensitivity is in need of a set of postulates
to ensure that future causal determinations are scientifically based. Below
is a set of postulates that, if met, would confirm (and if not met, refute)
that a person's symptoms were caused by a particular substance:
• When a subject simultaneously avoids all chemical, food and drug incitants,
remission of symptoms occurs (unmasking).
• A specific constellation of symptoms occurs with reintroduction of a
particular incitant.
• Symptoms resolve when the incitant is again avoided.
• With reexposure to the same incitant, the same constellation of symptoms
reoccurs, provided that the challenge is conducted within an appropriate window
of time.
Clinical observations suggest that an ideal window is four to seven days after
the last exposure to the test incitant.
To apply these postulates (illustrated in Figure 7), the timing of exposures
and the degree of masking should be rigorously controlled. To accomplish this,
a hospital-based clinical research facility, an Environmental Medical Unit
(EMU), is needed to isolate subjects from background exposures (Figure 8).4,5,15,16,26
The EMU would be constructed, furnished, and operated to minimize exposure
to airborne chemicals. For example, no disinfectants, perfumes, or pesticides
would be allowed in the unit. Ventilation would maximize fresh outside air
and provide optimal particulate and gas filtration. Patients would eat food
and water that is less chemically contaminated, testing one food per meal to
determine the effects of specific foods. If symptoms persisted despite this
approach, fasting for a few days would be attempted before reintroducing single
foods.
Figure 7. Graphic representation
depicting the testing of the toxicant-induced loss of tolerance postulates
using an environmental
medical unit.
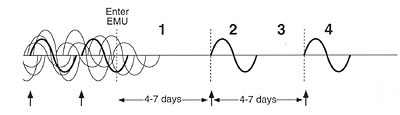
In the left-hand portion of the figure, a chemically sensitive
individual is experiencing symptoms in response to multiple exposures (chemicals,
foods, drugs) before entering the environmental medical unit. Effects overlap
in time. The effect of any particular exposure cannot be distinguished
from the effects of other exposures, and the person's symptoms may appear to
wax
and wane unpredictably over time. Postulate 1 – When all chemical, food,
and drug incitants are avoided concurrently, remission of symptoms occurs.
Anecdotally, patients report going through withdrawal or detox for the first
several days during which they experience symptoms such as increased irritability,
headaches, and depression. After 4 to 7 days, most report feeling well and
theoretically are at a clean baseline. Postulate 2 – A specific constellation
of symptoms occurs with reintroduction of an incitant. Postulate 3 – Symptoms
resolve when the incitant is again avoided. Postulate 4 – Reexposure
to the same incitant within an appropriate window of time (estimated to be
about 4–7 days) produces the same symptoms. For research purposes,
challenges should be conducted in a double-blind, placebo-controlled manner.
Figure
8. Preliminary design sketch of a patient
room in an environmental medical unit.
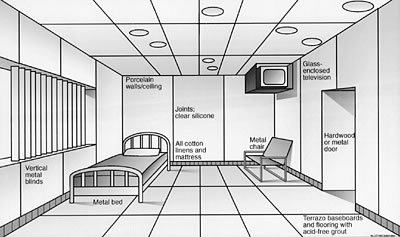
Note use of the non-outgassing construction
materials and furnishings and point source control (ventilated television
enclosure). Click here for a higher
quality image. (100
KB .pdf)
The rationale for housing subjects in an environmentally controlled facility
for several days before challenges is twofold: to prevent extraneous exposure
of patients to inhalants or ingestants so responses to them are not misinterpreted
as positive responses when placebo challenges are administered (false-positives),
and to minimize masking that might blunt or eliminate responses to active
challenges (false-negatives).
Although the terms exposure chamber and environmental medical unit appear
similar, conceptually they differ in important ways with regard to patient
safety and
control of interfering exposures.
By definition, an EMU is in a hospital where patients can remain 24 hours
a day in a clean environment for up to several weeks. Like an intensive
care
unit or coronary care unit, the EMU would be a specialized, dedicated hospital
facility. The EMU must be in a hospital to accommodate very sick patients;
exposure chambers do not offer comparable levels of care. Because chemical
challenges may precipitate bronchoconstriction, mental confusion, severe
headaches, depression, and other disabling symptoms, these patients should
not be tested
in an exposure chamber on an outpatient basis.
Conventional exposure chambers do not reduce background chemical exposures
for extended periods (up to several weeks), so the effects of a particular
challenge in a patient can be assessed accurately. This is the central
limitation of exposure chambers and the reason they should not be used
to rule in or rule
out chemical sensitivity. If subjects are not kept in a clean environment
for several days before and during challenges, false-positive responses
may occur
because of interfering exposures and false-negative responses may occur
because of masking. In contrast to an exposure chamber, an EMU would minimize
interfering
exposures before and during challenges, thus maximizing the reliability
and reproducibility of test responses.
Availability of an EMU would allow physicians to refer a wide variety of
cases in which environmental sensitivities were suspected to the unit for
definitive
evaluation. There, physicians could observe first-hand whether a patient's
symptoms improved after several days on a special diet in a clean environment.
If improvement occurred, single chemicals at concentrations encountered
in normal daily living, as well as single foods, could be reintroduced
one at
a time while the effects of each introduction were observed. Thus, the
EMU would be a tool to determine, in the most direct and definitive manner
possible,
whether chemical sensitivities exist. Studying patients with complicated
conditions like chronic fatigue syndrome or Gulf War syndrome in a conventional
exposure
chamber would not provide the same information, since chambers allow only
short-term residence, do not control the entire range of background contaminants,
and
provide inadequate separation from background exposures prior to challenges.
An analogy may help illustrate the importance of controlling exposures
for extended periods before challenge. If one wished to determine whether
a coffee
drinker's headaches were due to caffeine, it would not be adequate simply
to give the person a cup of coffee and ask him how he felt. It is obvious
that the individual would have to stop using caffeine for a period before
a meaningful
test of caffeine sensitivity could be performed. In this instance, a false-negative
challenge likely would be the result of failure to avoid coffee before
challenge. Similarly, placing a putatively sensitive person in a conventional
exposure
chamber and exposing him to a low concentration of a chemical might not
produce any noticeable effect. On the other hand, if this same person remained
in a
clean environment such as an EMU for a few days before being tested and
his condition improved, one could then perform meaningful challenges.
Placing patients in an EMU would simultaneously control all three components
of masking. Stopping all exposures several days before challenge testing
and spacing test exposures four to seven days apart would preclude acclimatization
or habituation. Eliminating background chemical noise and allowing the
effects
of each challenge to subside before introducing the next one would control
apposition. And excluding drugs, alcohol, nicotine, and caffeine while
spacing introduction of individual foods four to seven days apart would
interrupt any
addiction. Individual sensitivity could then be evaluated in the EMU, following
the postulates in Figure 7 for etiological verification.
For research purposes, challenges must be performed in a double-blind,
placebo-controlled manner. Patients with chronic fatigue syndrome, migraine
headaches, seizures,
depression, asthma, or unexplained illnesses such as Persian Gulf illness
could also be tested for sensitivities in an EMU using these postulates.
Thus, the
EMU could be used to determine whether particular patients with these diagnoses
had a masked form of this illness.
What evidence is there that unmasking patients in an EMU and conducting
challenges within a four- to seven-day window of time is either useful
or necessary? Thousands
of credible patients and dozens of physicians have attempted this approach.
They report that patients' symptoms resolve within a few days after they
enter such a facility and that robust symptoms occur when challenges are
conducted after several days of avoidance. Other evidence corroborates
these observations.
Withdrawal symptoms of several days' to a week's duration are known
to occur in some persons following cessation of exposure to nitroglycerine
(dynamite workers' headaches),27 caffeine,18,28 nicotine, and alcohol.
Note that these substances are chemically unrelated. In individuals chronically
exposed to xylene29 or ozone,30 reexposure after several days' avoidance
results in robust symptoms. Foods may require one to several days to navigate
the digestive tract before they are eliminated. Taken together, these observations
suggest that individuals with sensitivities to multiple incitants might
experience effects that linger as long as several days following initial
avoidance. Thus,
it may be argued that patients should be removed from their entire background
of food and chemical exposures for four to seven days before challenges,
as Randolph first proposed.14,17
While it is conceivable that synergistic or additive chemical combinations
may be necessary to reproduce certain symptoms, this is a limitation of
any form of challenge testing. Wherever possible within the bounds of safety
and
feasibility, chemical combinations believed to precipitate the most robust
and measurable responses should be explored. However, 40 years of clinical
observations, although anecdotal, suggest that single test substances may
suffice for most sensitive subjects. Confirmation or refutation of these
claims seems
a logical first step that should precede testing of complex mixtures. Finally,
because isolating patients in a hospital environment like the EMU may have
unanticipated psychological consequences, early studies in this area should
examine the responses of control subjects in the same environment.

Conclusion
Good pathological and physiological theories provide "a unified, clear,
and entirely intelligible meaning for a whole series of anatomical and clinical
facts, and for the relevant experiences and discoveries of reliable observers…."31
Theories and experiments that overlook salient observations or do not control
experimental conditions adequately may lead to erroneous conclusions. During
the late 19th century, researchers collected sputum from patients with tuberculosis
but were unsuccessful in culturing any organism. Some concluded that tuberculosis
was not an infectious disease. These early investigators did not know that
the tuberculin bacillus was fastidious and would grow out only after many weeks
on a specialized culture medium. Correspondingly, scientists' abilities
to observe and understand chemical sensitivity may depend on optimizing experimental
conditions, that is, appropriate timing of challenges and use of an EMU for
unmasking patients. To date, studies in this area have failed to unmask patients
before challenge. When false-positive and false-negative responses occurred,
investigators concluded that chemical sensitivity was psychogenic in origin.32,33
In summary, features of TILT overlap those of allergy, addiction, and classical
toxicity, yet TILT may be distinct from each of these. TILT appears to involve
a two-step process (resembling allergic sensitization) in which persons lose
specific tolerance (resembling addiction) for a wide range of common substances
following a chemical exposure event (resembling toxicity). Just as the germ
theory describes a class of diseases sharing the general mechanism of infection,
the TILT theory of disease posits a class of chemically induced disorders characterized
by loss of tolerance to chemicals, foods, drugs, and food and drug combinations.
In the same way that fever is a symptom commonly associated with infectious
diseases, chemical sensitivity may be a symptom associated with the TILT family
of diseases. Although clinical case definitions have been developed that describe
particular infectious diseases, no clinical case definition can be applied
to the entire class of infectious diseases. The same may be true for TILT disorders.
The fact that this phenomenon does not fit already accepted mechanisms for
disease is often offered as evidence that the condition does not exist. However,
the same criticism would have applied to the germ and immune theories of disease
when they first were proposed. What is plausible depends on the biological
knowledge of the time.34
Looking to the future, carefully conducted epidemiological studies and animal
models likely will play important roles in characterizing the initiation stage
of TILT during which tolerance is lost. In the meantime, rigorous testing of
the second stage of TILT, that is, the triggering of symptoms by tiny doses
of chemicals, foods, drugs, caffeine, or alcohol, is needed if progress in
this area is to occur. Adopting a set of relevant testable hypotheses for etiological
verification will ensure the credibility of those endeavors.

Claudia S. Miller, MD, MS
Associate Professor
Environmental and Occupational Medicine
Department of Family Practice
The University of Texas Health Science Center at San Antonio
7703 Floyd Curl Drive
San Antonio, Texas 78229-3900 USA
Website: www.uthscsa.edu

Reprinted from Environmental
Health Perspectives.
March 1997; Vol. 105, Supplement 2 and Townsend
Letter for Doctors and Patients. 210:76-84 (January 2001).
This paper is based on a presentation at the Conference on Experimental
Approaches to Chemical Sensitivity held 20-22 September 1995 in Princeton,
New Jersey. Research for this paper was supported in part by an appointment
to the Agency for Toxic Substances and Disease Registry (ATSDR) Clinical
Fellowship Program in Environmental Medicine, administered by Oak Ridge
Associated Universities through an interagency agreement between the
US Department of Energy and ATSDR.

Notes
1. Cullen MR, ed. Workers with multiple chemical sensitivities. Occup
Med: State Art Rev. 1987; 2(4):655–806.
2. Bascom R. Chemical Hypersensitivity Syndrome
Study: Options for Action, a Literature Review, and a Needs Assessment. A Report to the
State of Maryland Department of Environment. Baltimore, Maryland, 1989.
3. Ashford NA, Miller CS. Chemical Sensitivity.
A Report to the New Jersey State Department of Health. Trenton, NJ, 1989.
4. Ashford NA, Miller CS. Chemical Exposures:
Low Levels and High Stakes.
New York: Van Nostrand Reinhold, 1991.
5. National Research Council. Multiple Chemical
Sensitivities: Addendum to Biologic Markers in Immunotoxicology. Washington: National Academy
Press, 1992.
6. Association of Occupational and Environmental Clinics. Advancing
the understanding of multiple chemical sensitivity. Toxicol
Ind Health. 1992;8(4):1–257.
7. Thomson G. Report of the Ad Hoc Committee
on Environmental Hypersensitivity Disorders. Ontario, Canada, 1985.
8. Ashford N, Heinzow B, Lütjen K, Marouli C, Molhave L, Mönch
B, Papadopoulos S, Rest K, Rosdahl D, Siskos P, Velonakis E. Chemical
Sensitivity in Selected European Countries: An Exploratory Study. Athens:
Ergonomia, 1994.
9. Cone JE, Sult TA. Acquired intolerance to solvents following pesticide/solvent
exposure in a building: a new group of workers at risk for multiple
chemical sensitivities? Toxicol Ind Health. 1992;8(4):29–39.
10. Rosenthal N, Cameron CL. Exaggerated sensitivity to an organophosphate
pesticide (letter). Am J Psychiatry. 1991;148(2):270.
11. Ziem GE. Multiple chemical sensitivity: treatment and follow-up
with avoidance and control of chemical exposures. Advancing the understanding
of multiple chemical sensitivity. Toxicol
Ind Health. 1992;8(4):181–202.
12. Miller CS, Mitzel HC. Chemical sensitivity attributed to pesticide
exposure versus remodeling. Arch Environ Health. 1995;50(2):119–129.
13. Bell IR, Miller CS, Schwartz GE. An olfactory-limbic model of multiple
chemical sensitivity syndrome: possible relationships to kindling and
affective spectrum disorders. Biol Psychiatry. 1992;32:218–242.
14. Randolph TG, Moss RW. An Alternative Approach
to Allergies. New
York: Lippincott and Crowell, 1980.
15. Miller CS (1992). Possible models for multiple chemical sensitivity:
conceptual issues and role of the limbic system. Advancing the understanding
of multiple chemical sensitivity. Toxicol
Ind Health. 1992;8(4):181–202.
16. Miller CS. Chemical sensitivity: symptom, syndrome or mechanism
for disease? Toxicology. 1996;11:69–86.
17. Randolph TG. Human Ecology and Susceptibility
to the Chemical Environment. Springfield, IL: Charles C Thomas, 1962.
18. Silverman K, Evans SM, Strain EC, Griffiths RR. Withdrawal syndrome
after the double-blind cessation of caffeine consumption. N
Engl J Med. 1992;327(16):1109–1114.
19. Miller CS. Multiple chemical sensitivity and the Gulf War veterans.
Paper presented at The Persian Gulf Experience and Health, NIH Technology
Assessment Workshop, Bethesda, Maryland, 27–29 April 1994.
20. Waddell WJ. The science of toxicology and its relevance to MCS.
Reg Toxicol Pharmacol. 1993;18:13–22.
21. Fiedler N, Kipen HM, DeLuca J, Kelly-McNeil K, Natelson B. A controlled
comparison of multiple chemical sensitivities and chronic fatigue syndrome.
Psychosom Med. 1996;58:38–49.
22. Buchwald D, Garrity D. Comparison of patients with chronic fatigue
syndrome, fibromyalgia, and multiple chemical sensitivities. Arch
Intern Med. 1994;154:2049–2053.
23. Mølhave L, Bach B, Pederson OF. Human reactions to low concentrations
of volatile organic compounds. Environ Int. 1986;12:167–175.
24. AIHA. Odor Thresholds for Chemicals with
Established Occupational Health Standards. Fairfax, VA: American Industrial Hygiene Association,
1989.
25. Warner M. Hunting the yellow fever germ: the principle and practice
of etiological proof in late nineteenth-century America. Bull
Hist Med. 1985;59:361–382.
26. Miller CS. White paper: Chemical sensitivity: history and phenomenology.
Toxicol Ind Health. 1994;10(4/5):253–276.
27. Daum S. Nitroglycerin and alkyl nitrates. In: Environmental
and Occupational Medicine (Rom W, ed). Boston: Little Brown and Co, 1992;1013–1019.
28. Griffiths RR, Woodson PP. Caffeine physical dependence: a review
of human and laboratory animal studies. Psychopharmacology. 1988; 94:437–451.
29. Riihimaki V, Savolainen K. Human exposure to m-xylene. Kinetics
and acute effects on the central nervous system. Ann
Occup Hygiene.
1980;23:411–432.
30. Hackney JD, Linn WS, Mohler JG, Pedersen EE, Breisacher P, Russo
A. Experimental studies on human health effects of air pollutants.
Arch Environ Health. 1975;30:379–384.
31. Carter KC. Ignaz Semmelweiss, Carl Mayrhofer, and the rise of the
germ theory. Med Hist. 1985;29:33–53.
32. Leznoff A. Multiple chemical sensitivity: myth or reality? Prac
Allergy Immunol.. 1993;8(2):48–52.
33. Staudenmayer H, Selner JC, Buhr MP. Double-blind provocation chamber
challenges in 20 patients presenting with "multiple chemical
sensitivity." Reg Toxicol Pharmacol. 1993;18:44–53.
34. Hill AB. The environment and disease: association or causation?
Proc Royal Soc Med. 1965;58:295–300.
|



![]()
![]()







